 This is because the number of electrons determines chemical properties, and all three isotopes have one electron in their atoms. For example, carbon-12 is an isotope of carbon with a mass number of 12.Īll three isotopes of hydrogen have identical chemical properties. Chemical element, symbol Cl and atomic number 17 Chlorine, 17Cl Chlorine Pronunciation /klrin,-an/ (KLOR-een, -eyen) Appearance pale yellow-green gas Standard atomic weight Ar(Cl) 35.446, 35.457 35.450. 
IsotopeĪn isotope is named after the element and the mass number of its atoms. Hydrogen-1 is the most abundant (most common) isotope of hydrogen. Isotopes of an element can be easily spotted because:Īll hydrogen atoms contain one proton (and one electron because atoms must be electrically neutral), but they can contain different numbers of neutrons. 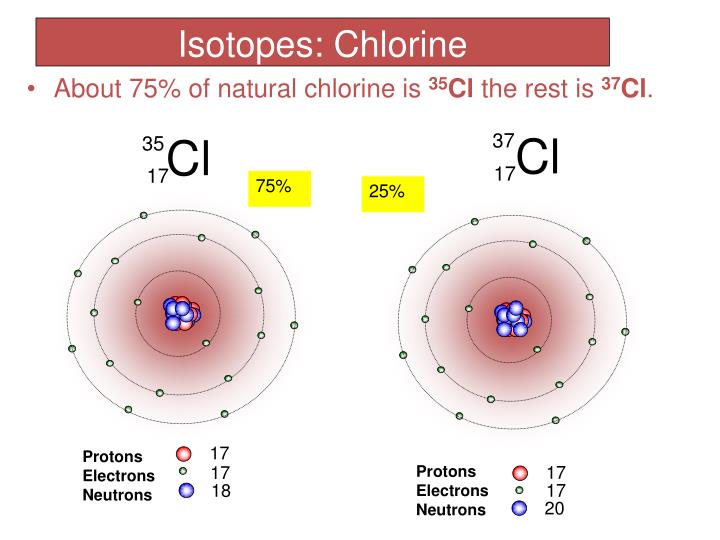
Atoms of an element that have the same number of protons, but different numbers of neutrons in their nuclei are called isotopes. It is known that the chlorine molecule is diatomic - Cl2, then the relative atomic mass of the chlorine molecule will be equal to: Ar (Cl2) 35,453 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
